 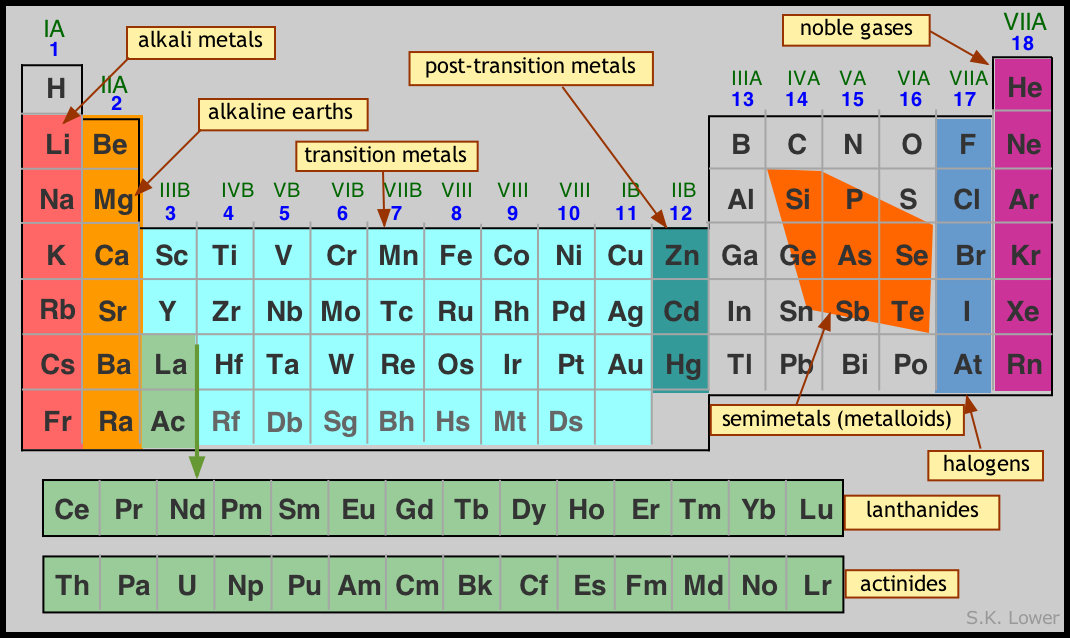
When the distance between these charged particles is larger, the electrostatic attraction between them becomes weaker. As the nucleus of an atom has an overall positive charge and electrons have a negative charge, there is an electrostatic attraction between them. Let’s now look at how this impacts the atoms’ properties. And as a result of this, the valence electron becomes further from the nucleus. So we know that the number of full electron shells increases as we descend the group. This pattern is repeated all the way down the group so that francium will have the most full electron shells. The number of full electron shells increases by one. The same is true when we go from sodium to potassium. So, as we go from lithium to sodium, the number of full electron shells increases by one. So a neutral atom of sodium will have 11 electrons: two in the first shell, eight in the second shell, and one in the outer shell. So a neutral atom of lithium will contain three electrons: two in the first shell and one in the second. And the size of the atoms increases as you descend the group. So they have one valence or outer shell electron. All of the alkali metals are in group one. Hydrogen is in group one of the periodic table, but it is not an alkali metal. 
The alkali metals include lithium, sodium, potassium, rubidium, cesium, and francium. The alkali metals can be found in group one of the periodic table. Which of the following alkali metals is the most reactive? (A) Cesium, (B) potassium, (C) lithium, (D) rubidium, or (E) sodium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
